UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 OR 15(d) of The Securities Exchange Act of 1934
Date
of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including
area code: (
(Former name or former address, if changed since last report.)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) |
Name of each
exchange on which registered | ||
| The |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
| Item 7.01 | Regulation FD Disclosure. |
On December 15, 2025, Palvella Therapeutics, Inc. (the “Company”) will host a conference call with investors at 8:30 a.m. Eastern Time, to present the topline results from the Company's Phase 2 TOIVA study of QTORIN™ 3.9% rapamycin anhydrous gel for the treatment of cutaneous venous malformations. The live event and accompanying slides can be accessed by visiting https://edge.media-server.com/mmc/p/cag4c7en/. A copy of the investor presentation is furnished herewith as Exhibit 99.1, and incorporated herein by reference.
The information furnished pursuant to Item 7.01, including Exhibit 99.1, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, and shall not be deemed to be incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as expressly set forth by specific reference in such filing.
| Item 8.01 | Other Events. |
On December 15, 2025, the Company issued a press release announcing positive topline results from the Company’s ongoing Phase 2 TOIVA study of QTORIN™ 3.9% rapamycin anhydrous gel for the treatment of cutaneous venous malformations. A copy of the press release is filed as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits
| Exhibit No. | Document | |
| 99.1 | Corporate Presentation on Phase 2 TOIVA Study of QTORIN™ dated December 15, 2025* | |
| 99.2 | Press Release issued by Palvella Therapeutics, Inc. on December 15, 2025 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
*Furnished herewith
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| PALVELLA THERAPEUTICS, INC. | |||
| Date: December 15, 2025 | By: | /s/ Matthew Korenberg | |
| Matthew Korenberg | |||
| Chief Financial Officer | |||
Exhibit 99.1

1 First - in - disease therapies for patients with rare skin diseases Phase 2 TOIVA Topline Data in Cutaneous Venous Malformations December 15, 2025

2 Forward Looking Statements This presentation contains forward - looking statements of Palvella Therapeutics, Inc. (“the Company”) within the meaning of the P rivate Securities Litigation Reform Act of 1995. Forward - looking statements include all statements that are not historical facts, and in some cases, can be identified by terms such a s “ may,” “might,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “objective,” “anticipate,” “believe,” “estimate,” “predict,” “potential,” “continue,” “ongoing,” or the neg ati ve of these terms, or other comparable terminology intended to identify statements about the future. Forward - looking statements contained in this presentation include, but are not limited to, statements regarding the Company’s future financial or business performance, conditions, plans, prospects, trends or strategies and other financial and business matters, the Compan y’s current and prospective product candidates and any additional indications or platform candidates, the Company's planned research and development activities, the Company's plan ned clinical trials, including timing of receipt of data from the same, the Company’s plan to meet with regulatory authorities, the planned regulatory framework for the Company's product cand ida tes including the Company’s plans to pursue Breakthrough Therapy Designation, the strength of the Company's intellectual property portfolio, and projections of the Compa ny’ s future financial results and other metrics. Such forward - looking statements are subject to risks, uncertainties, and other factors which could cause actual results to differ material ly from those expressed or implied by such forward looking statements. These forward - looking statements are based upon current estimates and assumptions of the Company and its management and are subj ect to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward - looking stat ements contained in this presentation. Factors that may cause actual results to differ materially from current expectations include, but are not limited to: competition, the ability of th e C ompany to grow and manage growth, maintain relationships with suppliers and retain its management and key employees; the success, cost and timing of the Company’s product development acti vit ies, studies and clinical trials; changes in applicable laws or regulations; the possibility that the Company may be adversely affected by other economic, business or competitive fa cto rs; the Company’s estimates of expenses and profitability; the evolution of the markets in which the Company competes; the ability of the Company to implement its strategic initiatives an d continue to innovate its existing products; and the ability of the Company to defend its intellectual property. Nothing in this Presentation should be regarded as a representation by any person that the forward - looking statements set forth herein will be achieved or that any of the contemplated results of such forward - looking statements will be achieved. You should not place undue reliance on forward - looking statements, which sp eak only as of the date they are made. The Company undertakes no duty to update these forward - looking statements. Industry and Market Data The Company may from time to time provide estimates, projections and other information concerning its industry, the general b usi ness environment, and the markets for certain conditions, including estimates regarding the potential size of those markets and the estimated incidence and prevalence of certain medic al conditions. Information that is based on estimates, forecasts, projections, market research or similar methodologies is inherently subject to uncertainties, and actual events, circumstance s o r numbers, including actual disease prevalence rates and market size, may differ materially from the information reflected in this presentation. Unless otherwise expressly stated, we ob tained this industry, business information, market data, prevalence information and other data from reports, research surveys, studies and similar data prepared by market research fi rms and other third parties, industry, medical and general publications, government data, and similar sources, in some cases applying our own assumptions and analysis that may, in the fut ure, prove not to have been accurate. Trademarks This Presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the propert y o f their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this Presentation may be listed without the TM, SM © or ® symbols, but the Company will assert, to the fullest extent under applicable law, the rights of the applicable owners, if any, to these trademarks, service marks, trade names and copyri ght s.

3 Phase 2 TOIVA Topline Data : Today’s Presenters Michael Kelly , MD, PhD • Pediatric hematologist - oncologist at Cleveland Clinic • Study investigator in Phase 2 TOIVA study • Member of Palvella Medical and Scientific Advisory Board Bohan Wei VP Corporate Development & New Product Planning Wes Kaupinen Founder and CEO Jeff Martini, PhD Chief Scientific Officer

4 Agenda EXECUTIVE SUMMARY DISEASE OVERVIEW PHASE 2 TOIVA TOPLINE RESULTS SUMMARY Q&A Wes Kaupinen, Founder and CEO Michael Kelly, MD, PhD, Cleveland Clinic Jeff Martini, PhD, CSO Michael Kelly, MD, PhD, Cleveland Clinic Wes Kaupinen, Founder and CEO

5 • Achieved statistical significance on multiple pre - specified clinician - reported and patient - reported efficacy endpoints, including dynamic change endpoints and static severity endpoints • Overall Cutaneous Venous Malformations - Investigators’ Global Assessment (cVM - IGA): 7 - point clinician - assessed change scale ranging from “Very Much Worse” ( - 3) to “Very Much Improved” (+3) • Mean effect size at week 12: +1.5 (p<0.001) • 73% of participants (11/15 participants) demonstrated 1 - point improvement or greater at Week 12 • 67% of participants (10/15 participants) rated as either “Much Improved” (+2) or “Very Much Improved” (+3) at Week 12 • QTORIN rapamycin was generally well - tolerated, consistent with previous clinical trials Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; Intent - to - Treat (ITT) population analyzed with no imputation of values for missing data. QTORIN Rapamycin for Cutaneous Venous Malformations: Phase 2 TOIVA Topline Data Planned discussions with FDA in early 2026 regarding the potential for Breakthrough Therapy Designation and a Phase 3 pivotal study

6 Dr. Michael Kelly, Cleveland Clinic Pediatric hematologist - oncologist at Cleveland Clinic • One of the leading physician - scientists advancing modern, mutation - informed treatment paradigms in the vascular anomalies field • Involved in over 100 clinical trials for novel oncology and vascular anomaly therapeutics • More than two decades of clinical and research experience in care of patients with complex venous, lymphatic, and mixed vascular malformations • Treated thousands of vascular anomaly patients, including those with cutaneous venous malformations and microcystic lymphatic malformations • Clinical investigator in Phase 2 TOIVA trial evaluating QTORIN rapamycin for the treatment of cutaneous venous malformations • Consultant to Palvella and investigator in the Phase 3 SELVA trial evaluating QTORIN rapamycin for microcystic lymphatic malformations Michael Kelly , MD, PhD

7 Disease Overview Dr. Michael Kelly, Cleveland Clinic QTORIN Rapamycin for Cutaneous Venous Malformations
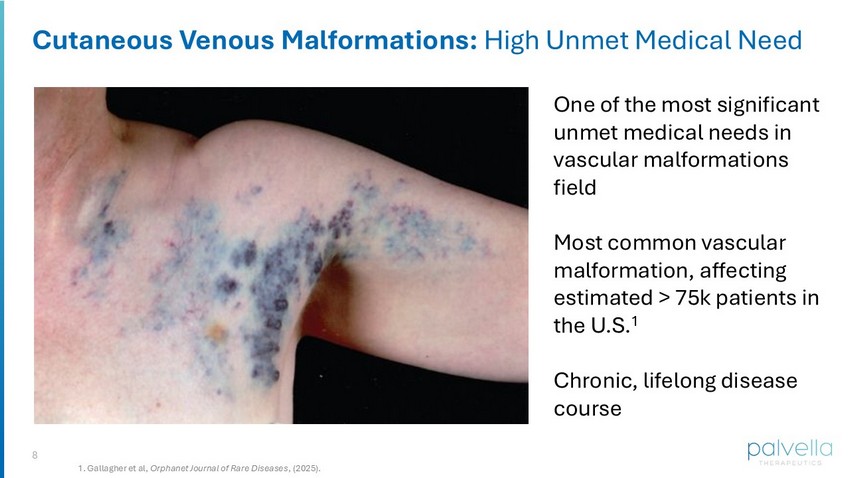
8 Cutaneous Venous Malformations: High Unmet Medical Need One of the most significant unmet medical needs in vascular malformations field Most common vascular malformation, affecting estimated > 75k patients in the U.S. 1 Chronic, lifelong disease course 1. Gallagher et al, Orphanet Journal of Rare Diseases , (2025).
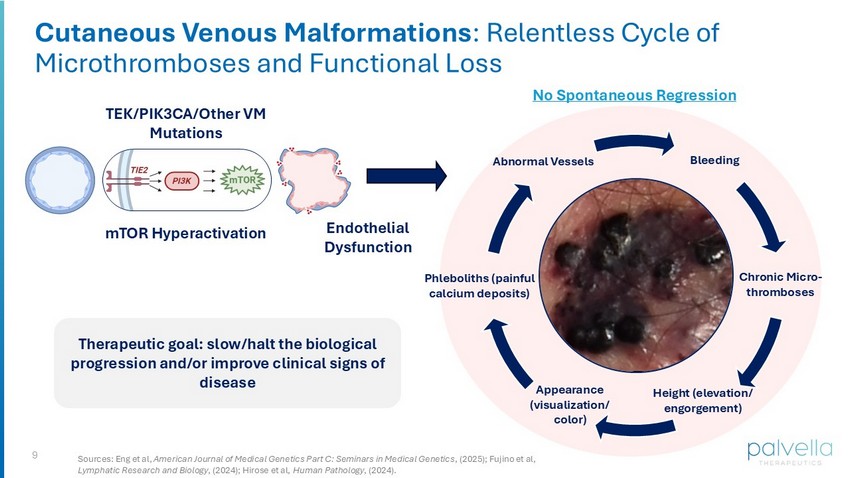
9 Cutaneous Venous Malformations : Relentless Cycle of Microthromboses and Functional Loss Abnormal Vessels Bleeding Chronic Micro - thromboses Height (elevation/ engorgement) Phleboliths (painful calcium deposits) TEK/PIK3CA/Other VM Mutations mTOR Hyperactivation Endothelial Dysfunction Therapeutic goal: slow/halt the biological progression and/or improve clinical signs of disease Appearance (visualization/ color) No Spontaneous Regression Sources: Eng et al, American Journal of Medical Genetics Part C: Seminars in Medical Genetics , (2025); Fujino et al, Lymphatic Research and Biology , (2024); Hirose et al, Human Pathology , (2024).

10 Constitutively Active Biological Signaling is Not Addressed by Non - specific, Episodic Treatments Sclerotherapy Laser Surgery ⮽ Does not address underlying causal biology of disease ⮽ Highly invasive and traumatic ⮽ Any improvement likely to be limited and short - term Disease will reliably recur, often more extensively than before Sources: Ali S, Mitchell SE Semin Intervent Radiol . 2017 Sep;34(3):288 - 293; Hassanein, AH, Plastic and Reconstructive Surgery 125(6):p 48, June 2010. Image Source: Vein Health Medical Clinic.

11 Phase 2 TOIVA Topline Results Dr. Jeff Martini, Chief Scientific Officer Dr. Michael Kelly, Cleveland Clinic QTORIN Rapamycin for Cutaneous Venous Malformations

12 Venous Malformations: Progress Towards the Potential First Targeted Therapy for Unaddressed Cutaneous Disease Known Genetics / Clear Biology Real - world Evidence Real - world evidence supports rapamycin’s off - label use in primarily internal manifestations of VMs… …however, poor patient outcomes persist in cutaneous disease Limaye et al (2009, 2015) Teng et al (2025) QTORIN RAPAMYCIN On Target, In Tissue Phase 2 TOIVA Data (2025) Plausible mechanism 3.9% concentration Dermal penetration Extensive CMC package QTORIN 3.9% rapamycin anhydrous gel is for investigational use only and has not been approved or cleared by the FDA or by any other regulatory agency. The safety or efficacy has not been established for any use.
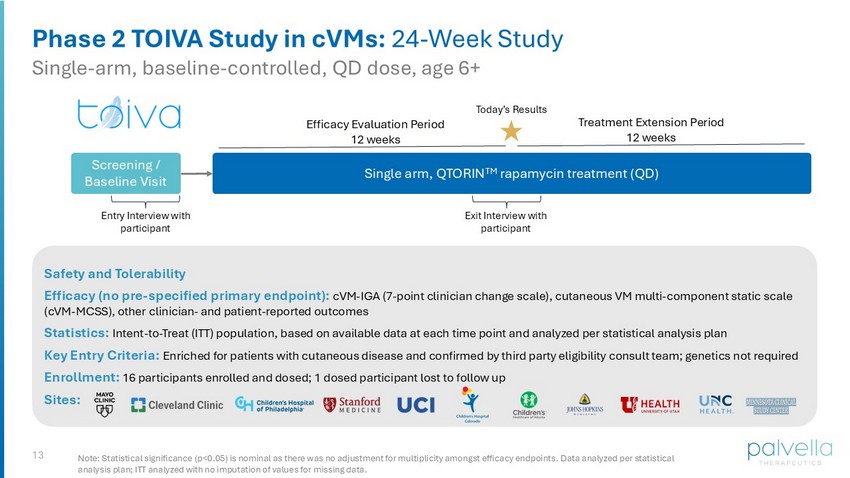
13 Phase 2 TOIVA Study in cVMs : 24 - Week Study Single - arm, baseline - controlled, QD dose, age 6+ Safety and Tolerability Efficacy (no pre - specified primary endpoint): cVM - IGA (7 - point clinician change scale), cutaneous VM multi - component static scale (cVM - MCSS), o ther clinician - and patient - reported outcomes Statistics: Intent - to - Treat (ITT) population, based on available data at each time point and analyzed per statistical analysis plan Key Entry Criteria: Enriched for patients with cutaneous disease and confirmed by third party eligibility consult team; genetics not required Enrollment: 16 participants enrolled and dosed; 1 dosed participant lost to follow up Sites: Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. Single arm, QTORIN TM rapamycin treatment (QD) Efficacy Evaluation Period 12 weeks Screening / Baseline Visit Exit Interview with participant Entry Interview with participant Treatment Extension Period 12 weeks Today’s Results
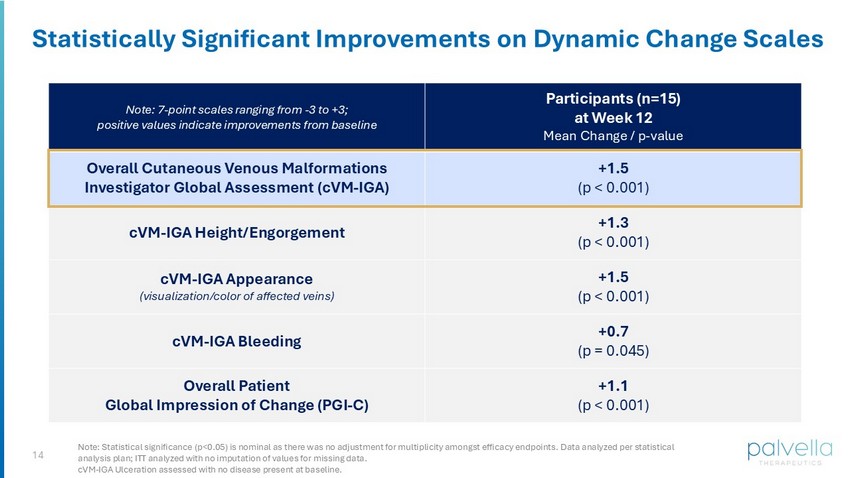
14 Statistically Significant Improvements on Dynamic Change Scales Participants (n=15) at Week 12 Mean Change / p - value Note: 7 - point scales ranging from - 3 to +3; positive values indicate improvements from baseline +1.5 (p < 0.001) Overall Cutaneous Venous Malformations Investigator Global Assessment (cVM - IGA) +1.3 (p < 0.001) cVM - IGA Height/Engorgement +1.5 (p < 0.001) cVM - IGA Appearance (visualization/color of affected veins) +0.7 (p = 0.045) cVM - IGA Bleeding +1.1 (p < 0.001) Overall Patient Global Impression of Change (PGI - C) Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. cVM - IGA Ulceration assessed with no disease present at baseline.
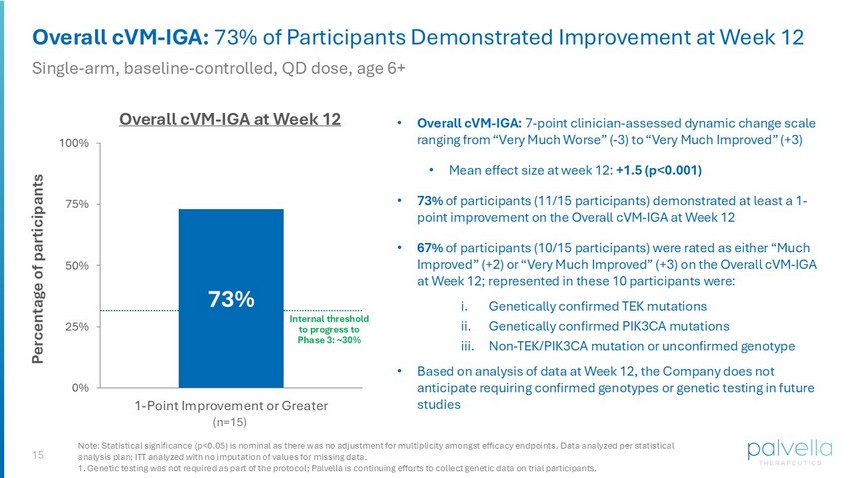
15 Overall cVM - IGA: 73% of Participants Demonstrated Improvement at Week 12 Single - arm, baseline - controlled, QD dose, age 6+ • Overall cVM - IGA: 7 - point clinician - assessed dynamic change scale ranging from “Very Much Worse” ( - 3) to “Very Much Improved” (+3) • Mean effect size at week 12: +1.5 (p<0.001) • 73% of participants (11/15 participants) demonstrated at least a 1 - point improvement on the Overall cVM - IGA at Week 12 • 67% of participants (10/15 participants) were rated as either “Much Improved” (+2) or “Very Much Improved” (+3) on the Overall cVM - IGA at Week 12; represented in these 10 participants were: i. Genetically confirmed TEK mutations ii. Genetically confirmed PIK3CA mutations iii. Non - TEK/PIK3CA mutation or unconfirmed genotype • Based on analysis of data at Week 12, the Company does not anticipate requiring confirmed genotypes or genetic testing in future studies Overall cVM - IGA at Week 12 (n=15) Internal threshold to progress to Phase 3: ~30% Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. 1. Genetic testing was not required as part of the protocol; Palvella is continuing efforts to collect genetic data on trial par ticipants. 73% 0% 25% 50% 75% 100% 1-Point Improvement or Greater Percentage of participants
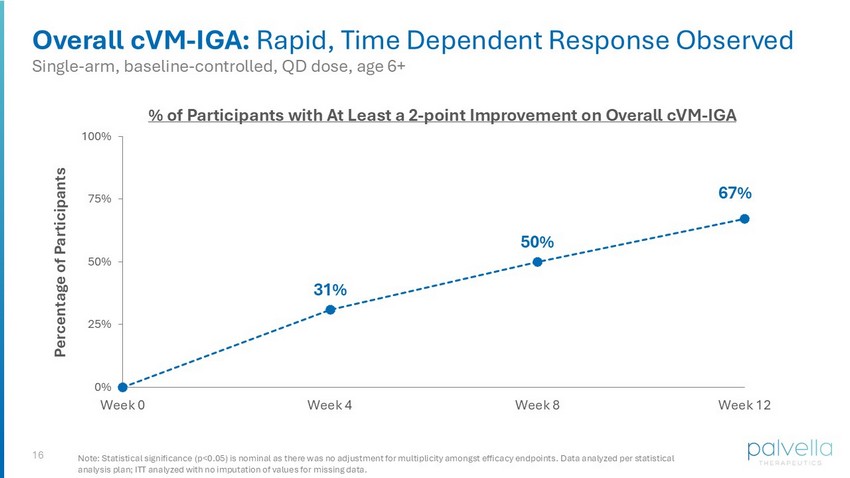
16 Overall cVM - IGA: Rapid, Time Dependent Response Observed % of Participants with At Least a 2 - point Improvement on Overall cVM - IGA 31% 50% 67% 0% 25% 50% 75% 100% Week 0 Week 4 Week 8 Week 12 Percentage of Participants Single - arm, baseline - controlled, QD dose, age 6+ Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data.

17 Statistically Significant Improvements on Key Static Severity Scales Participants (n=15) at Week 12 Mean Change / p - value Note: 5 - point scales ranging from 1 to 5; negative values indicate improvements from baseline - 1.0 (p < 0.001) Overall Clinician Global Impression of Severity (CGI - S) - 1.3 (p < 0.001) cVM - MCSS (Cutaneous VM Multi - Component Static Scale) Severity of Height/Engorgement - 1.1 (p < 0.001) cVM - MCSS Severity of Appearance (visualization/color of affected veins) - 0.5 (p = 0.027) Overall Patient Global Impression of Severity (PGI - S) Note: Statistical significance (p<0.05) is nominal as there was no adjustment for multiplicity amongst efficacy endpoints. Data analyzed per statistical analysis plan; ITT analyzed with no imputation of values for missing data. CGI - S Ulceration assessed with no disease present at baseline. CGI - S Bleeding assessed with limited disease present at baseline.
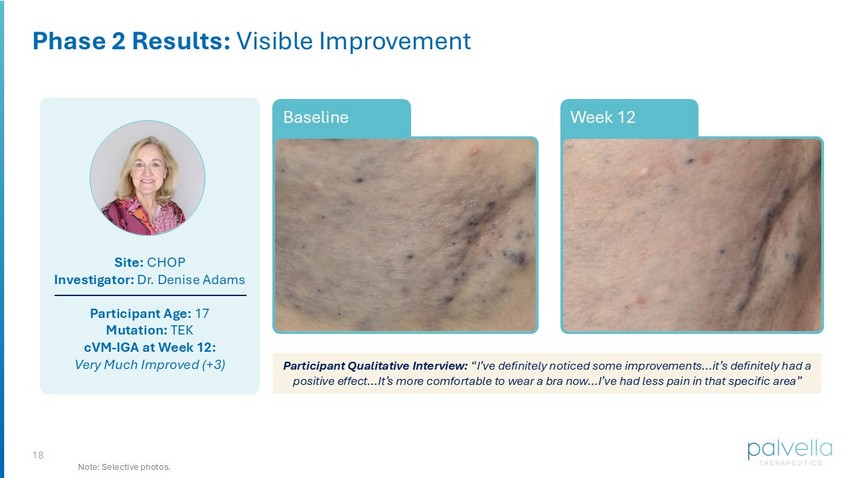
18 Site: CHOP Investigator: Dr. Denise Adams Participant Age: 17 Mutation: TEK cVM - IGA at Week 12: Very Much Improved (+3) Week 12 Baseline Phase 2 Results: Visible Improvement Participant Qualitative Interview: “I’ve definitely noticed some improvements…it’s definitely had a positive effect…It’s more comfortable to wear a bra now…I’ve had less pain in that specific area” Note: Selective photos.
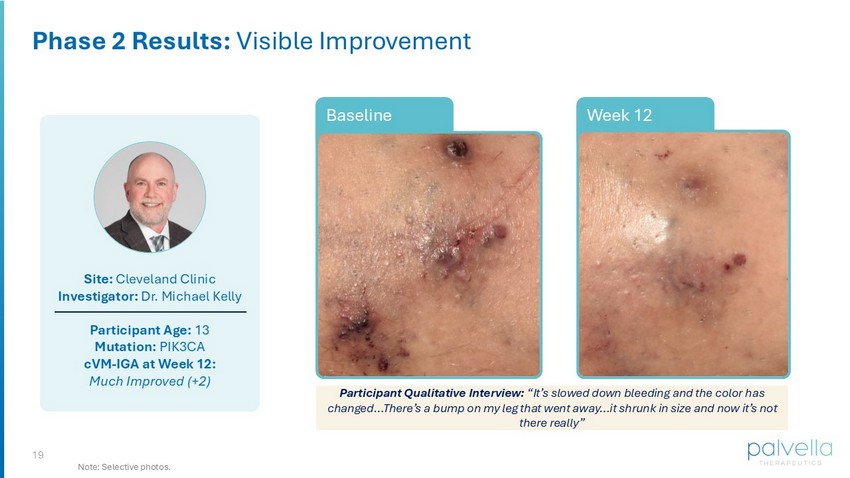
19 Site: Cleveland Clinic Investigator: Dr. Michael Kelly Participant Age: 13 Mutation: PIK3CA cVM - IGA at Week 12: Much Improved (+2) Week 12 Phase 2 Results: Visible Improvement Note: Selective photos. Week 12 Baseline Participant Qualitative Interview: “It’s slowed down bleeding and the color has changed…There’s a bump on my leg that went away…it shrunk in size and now it’s not there really”

20 Phase 2 Results: Safety and Tolerability • QTORI N rapamycin was generally well - tolerated , similar to previous clinical trials • Most common Treatment - Emergent Adverse Events were application site reactions (erythema, 25%) • All Treatment - Related Adverse Events were moderate or mild (no severe events) • Majority of AEs were mild • No SAEs related to study drug • No unexpected A Es Rapamycin levels were below the lower limit of quantification (2 ng/mL) in systemic circulation on a standard lab assay for all participants at all timepoints in the study Significantly below 5 ng/mL which is the lower boundary where rapamycin begins to exert immunosuppressive effects

21 Summary Wes Kaupinen Chief Executive Officer QTORIN Rapamycin for Cutaneous Venous Malformations

22 QTORIN Rapamycin has the Potential to Be the First FDA - Approved Therapy for Cutaneous Venous Malformations Achieved statistical significance on multiple pre - specified clinician - reported and patient - reported efficacy endpoints, including 73% of participants (11/15 participants) demonstrated a 1 - point improvement or greater on Overall cVM - IGA at Week 12 Planning for near - term discussions with FDA regarding potential for Breakthrough Therapy Designation and a Phase 3 pivotal study, as well as the newly announced Plausible Mechanism Pathway QTORIN rapamycin has the potential to become the first FDA - approved therapy and standard of care for the estimated more than 75,000 individuals with cutaneous venous malformations in the U.S. 1 The Phase 2 results further support Palvella’s “pipeline - in - a - product” strategy for QTORIN rapamycin, with programs advancing in microcystic LMs, cVMs, and angiokeratomas 2 3

23 Striving to be first for rare disease patients Thank You
Exhibit 99.2

Palvella Therapeutics Announces Positive Topline Results from Phase 2 TOIVA Clinical Trial of QTORIN™ 3.9% Rapamycin Anhydrous Gel (QTORIN™ rapamycin) for the Treatment of Cutaneous Venous Malformations, a Serious, Rare Genetic Disease with No FDA-approved Therapies
73% of trial participants (11/15 participants) improved on the Overall Cutaneous Venous Malformations Investigator Global Assessment (Overall cVM-IGA) at Week 12; 67% of trial participants (10/15 participants) rated as “Much Improved” (+2) or “Very Much Improved” (+3) on the Overall cVM-IGA at Week 12
Achieved statistical significance on multiple pre-specified clinician-reported and patient-reported efficacy endpoints, including dynamic change endpoints and static severity endpoints
QTORIN™ rapamycin was generally well-tolerated, with no drug-related serious adverse events reported
Based on Phase 2 results, Palvella to pursue near-term discussions with FDA regarding the potential for Breakthrough Therapy Designation and a Phase 3 pivotal study; FDA previously granted Fast Track Designation to QTORIN™ rapamycin for venous malformations
QTORIN™ rapamycin has the potential to become the first FDA-approved therapy and standard of care for the estimated more than 75,000 individuals with cutaneous venous malformations in the U.S.
Company to host conference call at 8:30am ET today
WAYNE, Pa., December 15, 2025 (GLOBE NEWSWIRE) — (Nasdaq: PVLA) Palvella Therapeutics, Inc. (Palvella or “the Company”), a clinical-stage biopharmaceutical company focused on developing and commercializing novel therapies to treat patients suffering from serious, rare skin diseases for which there are no U.S. Food and Drug Administration (FDA)-approved therapies, today announced positive topline results from the Company’s Phase 2 TOIVA study of QTORIN™ 3.9% rapamycin anhydrous gel (QTORIN™ rapamycin) for the treatment of cutaneous venous malformations (cutaneous VMs).
“Based on the large magnitude of the treatment effect observed in the majority of patients in the Phase 2 TOIVA study, QTORIN™ rapamycin has potential to become first-line therapy and to establish a much-needed standard of care for individuals living with cutaneous venous malformations,” said Megha Tollefson, M.D., pediatric dermatologist at Mayo Clinic and Principal Investigator of the Phase 2 TOIVA study. “Cutaneous venous malformations are congenital, chronic, progressive lesions that persist throughout life and can have a profound impact on patients’ quality of life. They may affect functionally critical areas of the body, often leading to daily discomfort, limitations in activities, and substantial burden for patients and their families. Current procedure-based approaches can be painful, ineffective, or both, and result in high rates of recurrence. Taken together, we believe the Phase 2 data represent a milestone day for individuals living with cutaneous venous malformations, and we look forward to working with Palvella to advance development of this therapy for patients as quickly as possible.”

TOIVA is a Phase 2, single-arm, open-label, baseline-controlled clinical trial of QTORIN™ rapamycin administered topically once daily for a 12-week efficacy evaluation period followed by a 12-week treatment extension period, for cutaneous VMs. The study enrolled 16 participants, ages six and older, at leading vascular anomaly centers across the U.S. Multiple measures of efficacy, including change in clinician and patient global impression assessments, as well as assessments of specific individual clinical manifestations which contribute to disease burden, were evaluated. To help contextualize changes on efficacy endpoints and, specifically, better understand any patient quality of life impact resulting from QTORIN™ rapamycin, qualitative exit interviews were conducted by a third-party interviewer with a subset of participants from the Phase 2 study.
Key findings from among the study's pre-specified efficacy endpoints at Week 12 demonstrated improvements in both clinician- and patient-reported outcomes:
| Efficacy Endpoints at Week 12 (ITT Population) | Mean Change from Baseline (n=15) | Nominal, Two-sided p-value | ||||||
| Dynamic Change Scales (7-point scales ranging from -3 to +3; positive values indicate improvements from baseline) | ||||||||
| Overall Cutaneous VM Investigator Global Assessment (Overall cVM-IGA) | 1.5 | <0.001 | ||||||
| cVM-IGA Height/Engorgement | 1.3 | <0.001 | ||||||
| cVM-IGA Appearance (visualization/color of affected veins) | 1.5 | <0.001 | ||||||
| cVM-IGA Bleeding | 0.7 | 0.045 | ||||||
| Overall Patient Global Impression of Change (PGI-C) | 1.1 | <0.001 | ||||||
| Static Severity Scales (5-point scales ranging from 1 to 5; negative values indicate improvements from baseline) | ||||||||
| Overall Clinician Global Impression of Severity (CGI-S) | -1.0 | <0.001 | ||||||
| cVM-MCSS (Cutaneous VM Multi-Component Static Scale) Severity of Height/Engorgement | -1.3 | <0.001 | ||||||
| cVM-MCSS Severity of Appearance (visualization/color of affected veins) | -1.1 | <0.001 | ||||||
| Overall Patient Global Impression of Severity (PGI-S) | -0.5 | 0.027 | ||||||
· p-values are nominal as there was no adjustment for multiplicity amongst efficacy endpoints; change scores and changes from baseline in static scores were compared vs. mean 0 using a 1-sample t-test.
· n=15 subjects completed treatment period (one additional dosed participant lost to follow up); data analyzed per statistical analysis plan; analysis conducted per available data at each timepoint; ITT analyzed with no imputation of values for missing data.
· Ulceration was also assessed with no disease present at baseline. CGI-S Bleeding assessed with limited disease present at baseline.
The Overall Cutaneous Venous Malformations Investigator Global Assessment (Overall cVM-IGA) is a 7-point, clinician-assessed, single-item efficacy endpoint measuring change in severity from baseline, with the numeric rating scale ranging from “Very Much Worse” (-3) to “Very Much Improved” (+3).
On the Overall cVM-IGA at Week 12:
| · | 73% of trial participants (11/15 participants) improved |
| · | 67% of trial participants (10/15 participants) were either “Much Improved” (+2) or “Very Much Improved” (+3) |
| · | No trial participants (0/15 participants) were “Minimally Worse” (-1), “Much Worse” (-2), or “Very Much Worse” (-3) |

Represented in the ten (10) participants who were rated either “Much Improved” (+2) or “Very Much Improved” (+3) on the Overall cVM-IGA at Week 12 were participants with (i) genetically confirmed TEK mutations, (ii) genetically confirmed PIK3CA mutations, and (iii) a non-TEK/PIK3CA mutation or unconfirmed genotype.
Genetic mutation confirmation was not required for entry into the Phase 2 TOIVA study. Additional data from trial participants, including mutation confirmation, continue to be collected, with genotype-stratified results planned for presentation at future medical meetings. Based on analysis of data at Week 12, the Company does not anticipate requiring confirmed genotypes or genetic testing in future studies.
Similar to previous clinical trials of QTORIN™ rapamycin, in the Phase 2 TOIVA study QTORIN™ rapamycin was generally well-tolerated, with the most common treatment-emergent adverse events being application site reactions (erythema, 25%). All treatment-related adverse events were moderate or mild, with no unexpected adverse events reported. Rapamycin levels were below the lower limit of quantification (2 ng/mL) in systemic circulation on a standard lab assay for all participants at all timepoints in the study.
"Based on the strength of the Phase 2 TOIVA results, including the 73% of participants who improved on the Overall cVM-IGA at Week 12, Palvella is planning for near-term discussions with FDA regarding the potential for Breakthrough Therapy Designation and a Phase 3 pivotal study, as well as the newly announced Plausible Mechanism Pathway," said Wes Kaupinen, Founder and Chief Executive Officer of Palvella. “We are deeply grateful to the patients and families who participated in this trial, as well as the investigators and clinical centers whose partnership made this study possible. These data reinforce our conviction in the therapeutic potential of QTORIN™ rapamycin, the strength of the QTORIN™ platform, and our long-term vision to become the leading rare disease company focused on developing and commercializing novel therapies for patients with serious, rare skin diseases.”
FDA has previously granted Fast Track Designation to QTORIN™ rapamycin for the treatment of venous malformations.
Conference Call Details
Palvella will host a conference call and live audiovisual webcast to discuss the Phase 2 TOIVA topline results at 8:30 a.m. ET today. To access the live webcast of the call with slides, please click here or visit the "Events & Presentations" section of Palvella’s website. To access the call by phone, please use this registration link, and you will be provided with dial in details. A replay of the webcast will be available approximately 2 hours after the conclusion of the call and archived for 90 days under the "Events & Presentations" section of the Company's website at www.palvellatx.com.

About Cutaneous Venous Malformations
Cutaneous VMs are a rare genetic disease caused by mutations in genes that cause overactivation of the PI3K/mTOR signaling pathway, leading to dysfunctional veins within the skin. These malformations can cause substantial morbidity and functional impairment, significantly impact quality of life, and are associated with severe bleeding, thrombosis, and other potential complications. An urgent need exists for an FDA-approved, targeted, localized therapy to treat cutaneous VMs. While published case studies and real-world evidence have provided preliminary evidence of clinical benefit from the off-label use of systemic mTOR inhibitors for venous malformations, there are currently no FDA-approved therapies for the estimated more than 75,000 diagnosed patients with cutaneous VMs in the U.S.
About Palvella Therapeutics
Founded and led by rare disease drug development veterans, Palvella Therapeutics, Inc. (Nasdaq: PVLA) is a clinical-stage biopharmaceutical company focused on developing and commercializing novel therapies to treat patients suffering from serious, rare skin diseases for which there are no FDA-approved therapies. Palvella is developing a broad pipeline of product candidates based on its patented QTORIN™ platform, with an initial focus on serious, rare skin diseases, many of which are lifelong in nature. Palvella’s lead product candidate, QTORIN™ 3.9% rapamycin anhydrous gel (QTORIN™ rapamycin), is currently being developed for the treatment of microcystic lymphatic malformations, cutaneous venous malformations, and clinically significant angiokeratomas. Palvella’s second product candidate, QTORIN™ pitavastatin, is currently being developed for the topical treatment of disseminated superficial actinic porokeratosis. For more information, please visit www.palvellatx.com or follow Palvella on LinkedIn or X (formerly known as Twitter).
QTORIN™ rapamycin and QTORIN™ pitavastatin are for investigational use only and neither has been approved by the FDA or by any other regulatory agency for any indication.
Forward-Looking Statements
This press release contains forward-looking statements (including within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended (Securities Act)). These statements may discuss goals, intentions, and expectations as to future plans, trends, events, results of operations or financial condition, or otherwise, based on current beliefs of the management of Palvella, as well as assumptions made by, and information currently available to, the management of Palvella. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as “may,” “will,” “should,” “would,” “expect,” “anticipate,” “plan,” “likely,” “believe,” “estimate,” “project,” “intend,” and other similar expressions or the negative or plural of these words, or other similar expressions that are predictions or indicate future events or prospects, although not all forward-looking statements contain these words. Statements that are not historical facts are forward-looking statements. Forward-looking statements include, but are not limited to, statements regarding the expected timing of the presentation of data from ongoing clinical trials, including the TOIVA study, Palvella’s clinical development plans and related anticipated development milestones, Palvella’s plans to pursue Breakthrough Therapy Designation, Palvella’s plans to meet with regulatory authorities, Palvella’s cash, financial resources and expected runway, Palvella’s expectations regarding its programs, including QTORIN™ rapamycin and QTORIN™ pitavastatin, and its research-stage opportunities, including its expected therapeutic potential and market opportunity. Forward-looking statements are based on current beliefs and assumptions that are subject to risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the ability to raise additional capital to finance operations; the ability to advance product candidates through preclinical and clinical development; the ability to obtain regulatory approval for, and ultimately commercialize, Palvella’s product candidates, including QTORIN™ rapamycin and QTORIN™ pitavastatin; the outcome of early clinical trials for Palvella’s product candidates, including the ability of those trials to satisfy relevant governmental or regulatory requirements; the fact that data and results from clinical studies may not necessarily be indicative of future results; Palvella’s limited experience in designing clinical trials and lack of experience in conducting clinical trials; the ability to identify and pivot to other programs, product candidates, or indications that may be more profitable or successful than Palvella’s current product candidates; the substantial competition Palvella faces in discovering, developing, or commercializing products; the negative impacts of global events on operations, including ongoing and planned clinical trials and ongoing and planned preclinical studies; the ability to attract, hire, and retain skilled executive officers and employees; the ability of Palvella to protect its intellectual property and proprietary technologies; reliance on third parties, contract manufacturers, and contract research organizations; and the risks and uncertainties described in the filings made by Palvella with the Securities and Exchange Commission (SEC), including the annual report on Form 10-K, quarterly reports on Form 10-Q and current reports on Form 8-K, filed with or furnished to the SEC and available at www.sec.gov. The events and circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that Palvella may face. Except as required by applicable law, Palvella does not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. This press release contains hyperlinks to information that is not deemed to be incorporated by reference into this press release.

Contact Information
Investors
Wesley H. Kaupinen
Founder and CEO, Palvella Therapeutics
wes.kaupinen@palvellatx.com
Media
Marcy Nanus
Managing Partner, Trilon Advisors LLC
mnanus@trilonadvisors.com
